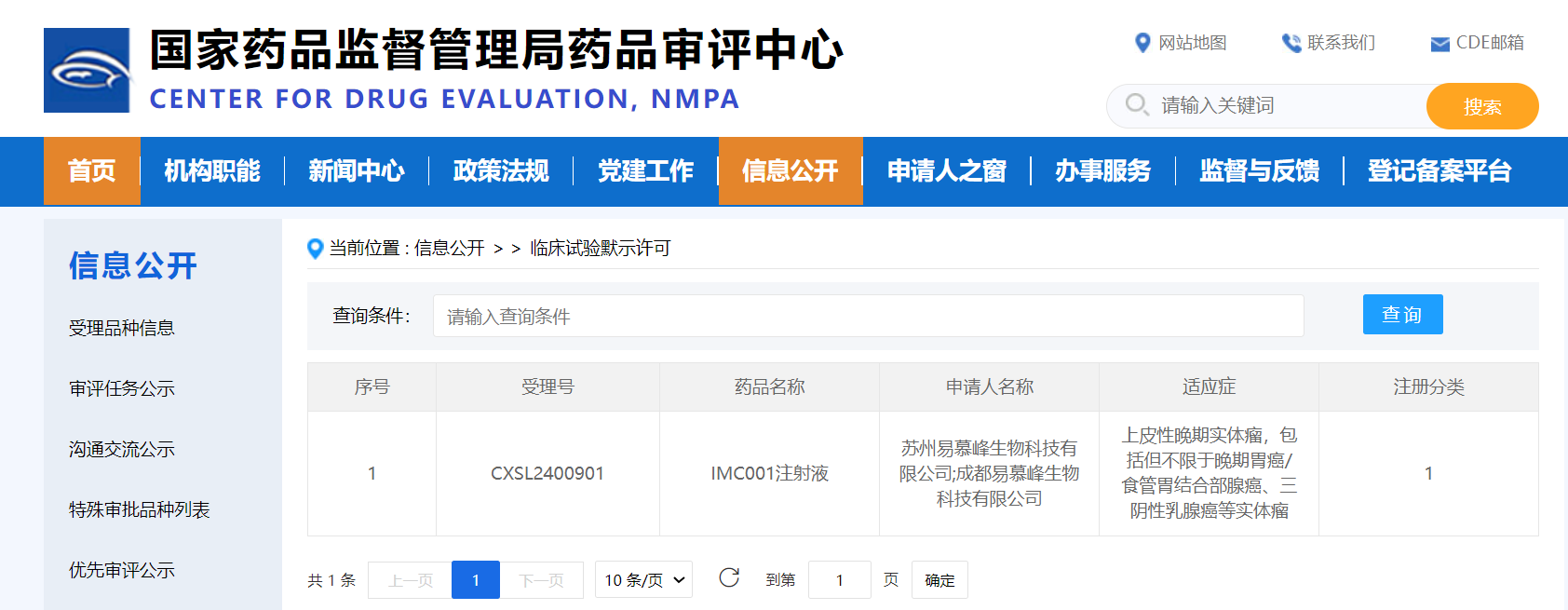
On March 21, 2025,Immunofoco announced that its independently developed EpCAM-targeting autologous CAR-T cell therapy, IMC001, has been granted clearance by the Center for Drug Evaluation (CDE) under the National Medical Products Administration (NMPA) in China for its second Investigational New Drug (IND) application, with the acceptance number CXSL2400901. This IND application is aimed at treating EpCAM-positive epithelial advanced solid tumors. Previously, IMC001 had obtained IND approvals in both China and the United States in February 2024 for the treatment of EpCAM-positive advanced gastrointestinal tumors.
Epithelial cell adhesion molecule (EpCAM) is highly expressed on various epithelial-derived tumor tissues, including esophageal cancer, colorectal cancer, pancreatic cancer, ovarian cancer, lung cancer, gastric cancer, and breast cancer. In contrast, its expression is relatively low in normal tissues. Despite challenges such as the complexity of the solid tumor microenvironment and target heterogeneity, IMC001 has demonstrated favorable safety and preliminary efficacy in earlier clinical trials for gastrointestinal tumors, owing to its unique target selection and innovative design. This additional indication aims to further explore its clinical potential.
In the poster presentations at the 2024 ASCO and CSCO conferences, the IIT clinical study data of IMC001 demonstrated that among 10 evaluable patients with advanced gastric cancer, the disease control rate was 90%, with one patient in the low-dose group 33.3% and two in the middle-dose group 40% achieved a partial response (PR). The median overall survival (OS) in the medium-dose group exceeded 55 weeks. One patient in the middle-dose group achieved a confirmed PR by Week 24, leading to a radical gastrectomy at Week 27, and the patient had survived for more than 22 months by the cutoff date.
The clearance of this additional IND represents a pivotal milestone in expanding IMC001’s clinical exploration scope from gastrointestinal tumors to a broader spectrum of epithelial solid tumors, including small-cell lung cancer (SCLC), triple-negative breast cancer (TNBC), and other malignancies.
At present, there are still many clinical trials of new anti-cancer technologies in China seeking patients. Consultation on new drugs and technologies, you can contact Beijing South Region Oncology Hospital International Department.
Phone Number:4008803716
Email:myimmnet@163.com
Post time: Apr-03-2025

